Histologic and Histomorphometric Evaluation of Implant Osseointegration of a Dental Implant Three Years in Function Removed Due to Abutment Fracture
Abstract
For the edentulous patient, dental implant therapy is the accepted treatment to replace one or more missing teeth as implant survival rates are greater than 90%. Unfortunately, surgical removal of dental implants occurs for a variety of reasons. As human studies are limited, this case report provides an opportunity to evaluate the peri-implant characteristics in a dental implant removed from a patient using histology. In our patient, the implant was removed three years after completion of the prosthetic phase due to fracture of the abutment that could not be removed from the implant. Histological examination of the implant revealed mature lamellar bone in direct contact with the implant surface. Histomorphometric evaluation revealed a bone-to-implant contact (BIC) of 73.6%.
Author Contributions
Academic Editor: Sasho Stoleski, Institute of Occupational Health of R. Macedonia, WHO CC and Ga2len CC
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2026 Cameron Y. S. Lee, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors declare no conflicts of interest.
Citation:
Introduction
For the partially or totally edentulous patient, dental implant treatment is the accepted and preferred treatment modality 1, 2 as long-term implant survival rates are greater than 90% 3, 1. Implant osseointegration is dependent on the physicochemical characteristics of the implant surface as direct bone-to-implant contact (BIC) at the peri-implant interface is crucial 4. Therefore, BIC has proven to be a critical histomorphometric parameter for successful osseointegration and is highly influenced by implant surface modifications as they affect osteoblast activity and osteogenesis 5, 6.
Implant surface modifications have improved osseointegration since Branemark introduced machined titanium dental implants 7. Because titanium is a low bioactivity material, biomaterials research has concentrated on implant surface modifications to increase osseointegration 8. One strategy to improve implant osseointegration is to coat the implant surface with hydroxyapatite (HA) because of its bioactivity, biocompatibility and osteoconductive properties 9. HA is a non-toxic material, does not elicit an inflammatory response and is non-immunogenic 10. HA provides an osteophilic surface that increases BIC due to its osteoinductive properties 11.
Despite long-term implant survival rates greater than 90% 3, 1 implants will need to be removed for a variety of reasons. Biological problems that lead to implant removal include periimplantitis, loss of osseointegration, and fracture of the implant body. Prosthetic problems include an unrestorable implant due to poor alignment, and abutment fracture 12. However, an abutment fracture provides an opportunity to study the peri-implant histology and bone-to-implant contact (BIC). In this case report, the authors had the opportunity to remove the implant after three years in function and examine the histology of implant osseointegration with the surrounding bone.
Materials and Methods
With fracture of the prosthetic abutment, it is often difficult to remove the abutment shaft from the internal well of the implant. In this situation, the implant cannot be restored and must be removed if the treatment plan is to place a new implant to restore patient function. BIC histologic analysis in humans provides the clinician with a greater understanding of implant osseointegration 13, 14 as most studies on BIC have relied on animal studies 12.
Case Report
The patient is a 56-year-old Asian female who presented to the office with the chief complaint that her “tooth is loose” when chewing her food. In February 2022, the patient completed implant surgery with a screw-type implant with the sinus lift elevation with bone grafting procedure to replace missing tooth #03. The post-operative course was unremarkable, and the prosthetic phase was completed four months after implant surgery. In December 2025, examination of the right posterior maxilla revealed the crown missing from the implant. Closer inspection demonstrated fracture of the prosthetic abutment at the coronal part of the implant. Attempts to remove the fractured abutment from the well of the implant were not successful. The decision was made to replace the existing implant from the posterior maxilla and surgically place a new implant. Informed consent was obtained from the patient. The implant and surrounding bone were removed using a trephine bur under cool water irrigation. The surgical site was bone grafted in preparation for future implant surgery. After removal of the implant from the posterior maxilla, the implant was placed in 10% neutral buffered formalin. The implant was sent to the Hard Tissue Research Laboratory at the University of Minnesota School of Dentistry for histological examination of the implant and surrounding bone.
Specimen Processing
The implant with its core of bone attached to it were sectioned in half buccolingually and immediately dehydrated with a graded series of ethanol for 9 days. After dehydration, each specimen was infiltrated for 20 days with a light-curing embedding resin (Technovit 7200 VLC, Kulzer, Wehrheim, Germany). The implant was then embedded in Technovit 7200 VLC and polymerized by 450-nm light, with the temperature of the specimens not exceeding 40 degrees centigrade. The specimens were prepared by the cutting and grinding method of Rohrer and Schubert 15. Each specimen was cut to a thickness of 150 mm on an EXAKT cutting and grinding system (EXAKT Technologies, Oklahoma City, Okla). Each specimen slide was then polished to a thickness of 35 um with a series of polishing sandpaper discs ranging from 800 to 2400 grit (EXAKT micro grinding system) followed by a final polish with 0.3 um alumina polishing paste. This thickness is ideal for the preservation of titanium on the microscope slide and high-power light microscope evaluation of undecalcified bone.
After final polishing, the specimen slides were stained using Stevenel’s blue and van Gieson’s picro fuchsin for histological evaluation by light microscopy. The implant was evaluated using two slides to prevent sampling bias. Microphotographs were obtained, scanned, digitized, and analyzed using a Zeiss Axiolab photomicroscope (Carl Zeiss, Jena, Germany) and Nikon Coolpix 4500 digital camera (Nikon Corp, Tokyo, Japan). The core specimen was photographed at a fixed focal point at 25x magnification for histomorphometric evaluation. Histomorphometric measurements were completed with a Macintosh G4 computer (Apple, Cupertino, Calif) and a public domain image program. NIH Images, US National Institutes of Health) along with Adobe Photoshop (Adobe, San Jose, Calif). The data were exported to Microsoft Excel (Microsoft Co, Redmond, Wash) for histomorphometric calculations. Histomorphometric analysis was performed, and the following parameters were measured in terms of the percentage of the total core area: new bone formation, residual graft material, and marrow spaces. In addition, using circularly polarized light birefringence was evaluated for transverse collagen fiber orientation.
Results
The implant was osseointegrated with a BIC of 73.6%. No dissolution or resorption of the hydroxyapatite coating was observed (Figure 3). The surrounding bone was in direct contact with the hydroxyapatite coating of the implant (Figure 1, Figure 2, Figure 3, Figure 4). Histologic examination revealed mineralized mature cortical bone in direct contact along the implant surface (Figure 1, Figure 2 and Figure 3). Lamellar cortical bone was observed in the vent area at the apical part of the implant (Figure 1). These findings reveal long-term implant osseointegration.
Figure 1.Low power view. Retrieved dental implant. Implant threads in direct contact with mature lamellar bone (Original magnification x 20). Note mature vital bone in apical vent of implant.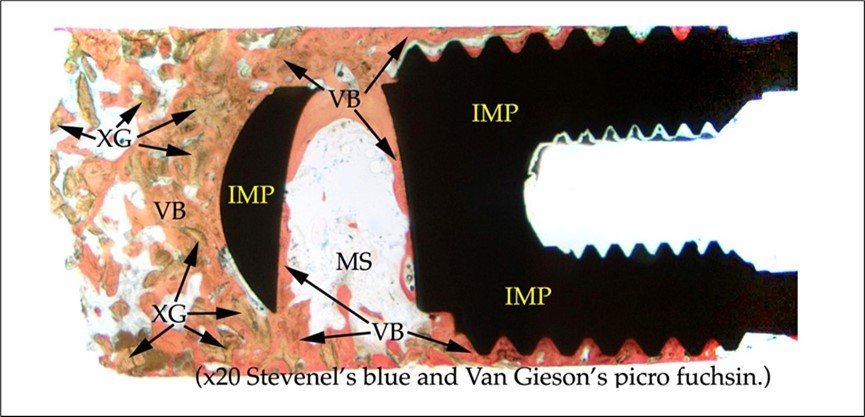
Figure 2.Medium power view. Bone core in apical area of implant. A. Light microscopy showing marrow spaces and particles of xenograft bridging with mature autogenous vital bone (Original magnification x 40). B. Circularly polarized light microscopy showing orientation of different groups of bone formation (Original magnification x 20).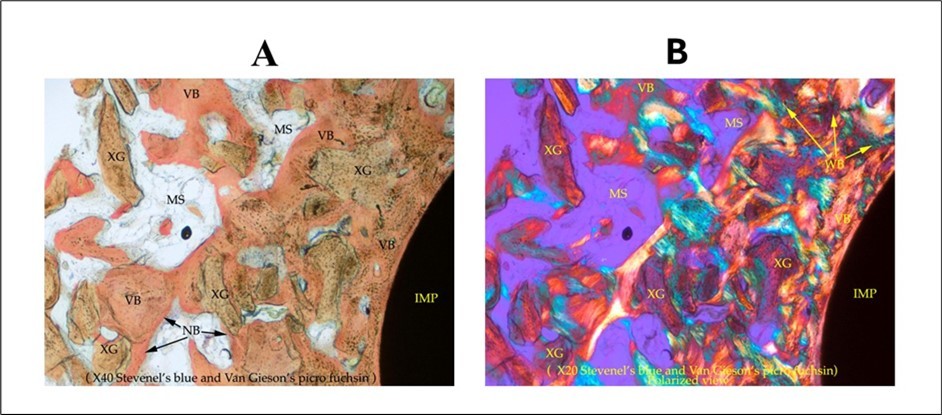
Figure 3.High power view. Mature vital bone in direct contact with implant surface (Original magnification x 40).
Figure 4.High power view. Hydroxyapatite coating of implant in direct contact with surrounding bone and the large number of vital osteocytes in their lacunae and osteoblasts on the implant surface. Bone has infiltrated into the HA surface and no dissolution or resorption of HA coating observed (Original magnification x 200).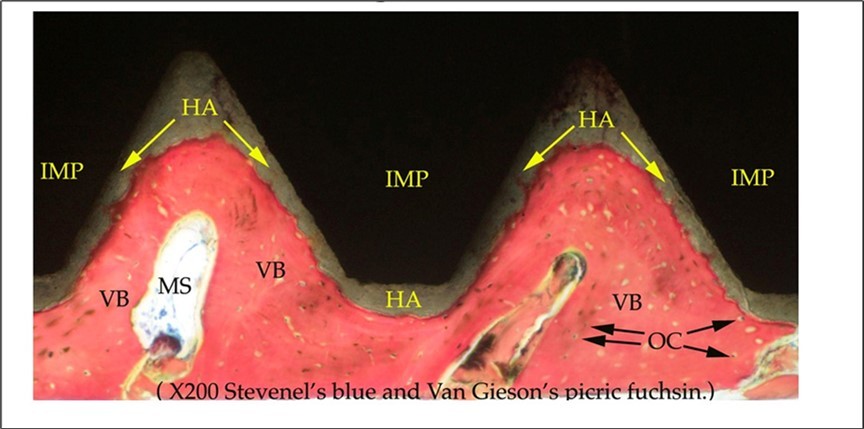
Discussion
Removal of dental implants provides the opportunity for the clinician to obtain valuable information on implant osseointegration during different healing time periods 16. In a study by Tumedei et al. (2020), the two most frequent reasons for removal of implants were abutment fracture in 65% of prosthetic cases and implant fracture in 25%. Although human studies are few, most of the histological studies reported in the literature on implants removed showed compact, mature lamellar bone with haversian canals and osteocytes 17, 18, 19, 20. Our case report also showed mature lamellar bone with osteocytes in their lacunae in direct contact with the implant surface (Figure 4). No fibrous tissue was observed.
Using a surface modified screw-type implant increases the surface area and osseointegration 21. Mature lamellar bone, haversian canals and osteocytes were observed histologically in Figure 1, Figure 2, Figure 3, Figure 4. At high power view, the HA implant surface coating was well integrated at the peri-implant surface. Histologic findings were consistent with studies by Piattelli et al. 22, Molly et al. 23 and Marco et al. 24. All three studies reported no significant inflammatory response when using a xenograft substitute bone graft material. In our patient, after three years in function no inflammatory response was observed.
In a study by Faeda et al. (2019), three fractured dental implants were removed from the maxilla in three different patients. Histomorphometric analysis revealed a mean bone-to-implant contact of 77.3%. Piattelli et al. (1997) reported a mean BIC of 60 to 70% with a titanium plasma sprayed implant removed from a patient. Brunel et al. (2020) reported a 74% BIC in a hydroxyapatite coated implant removed from the maxilla after 14 months of follow-up. In our case report, a bone-to-implant contact of 73.6% was observed.
Conclusion
This case report examines the histology of long-term osseointegration at the bone-implant interface in a human patient. Histology demonstrated the implant surrounded by mature vital bone. Histomorphometric evaluation showed a bone-to-implant contact percentage of 73.6%. Results of this case report are similar to published articles in the implant literature.
Declaration
The authors declare that they have no financial interest in the publication of this case report.
Abbreviations
References
- 1.Lekholm U, Gunne J, Henry P. (1999) Survival of the Branemark implant in partially edentulous jaws: a 10-year prospective study. , Int J Oral Maxillofac Implants 14, 639-645.
- 2.Howe M S, Keys W, Richards D. (2019) Long-term (10-year) dental implant survival: a systematic review and sensitivity analysis. , Dent, J 84, 9-21.
- 3.Branemark P I, Hansson B O, Adell R. (1977) Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scan J Plast Reconstr Surg. 16-1.
- 5.DMD Ehrenfest, Coelho P G, Kang B S. (2010) Classification of osseointegrated implant surfaces: materials, chemistry and topography. Trends Biotechnol. 28(4), 198-206.
- 6.Smeets R, Stadlinger B, Schwarz F. (2016) Impact of dental implant surface modifications on osseointegration. Biomed Res Int. 2016(1), 6285620-10.
- 7.Branemark P I, Adell R, Breine U. (1969) Intra-osseous anchorage of dental prostheses. , I. Experimental 3, 81-100.
- 8.Palmquist A, Omar M, Esposito M. (2010) Titanium oral implants: Surface characteristics, interface biology and clinical. 7, 515-527.
- 9.T-MG Chu, Orton D G, Hollister S J. (2002) Mechanical and in vivo performance of hydroxyapatite implants with controlled architecture. Biomaterials. 23(5), 1283-1293.
- 10.Qadir M, Li Y, Wen C. (2019) Ion-substituted calcium phosphate coatings by physical vapor deposition magnetron sputtering for biomedical applications: A review. Acta Biomater. 89, 14-32.
- 11.López-Valverde N, Flores-Fraile J, Ramírez J M. (2020) Bioactive Surfaces vs. Conventional Surfaces in Titanium Dental Implants: A Comparative Systematic Review. , J Clin Med 9(7), 1-26.
- 12.THI Skiba, Kalil E C, Piattelli A. (2023) Human histological analysis of early bone response to immediately loaded narrow dental implants with biphasic calcium phosphate grid blasted surface treatment: a case report. , Dent J 11, 177-184.
- 13.Coelho P G, Bonfante E A, Marin C. (2010) A human retrieval study of plasma-sprayed hydroxyapatite-coated plateau root form implants after 2 months to 13 years in function. , J. Long. Term. Eff. Med. Implants 20, 335-342.
- 14.Abbott J R, Marino V, Bartold P M. (2014) Human cadaveric histomorphological and metallurgical analysis of dental implants following 12.5 years of service. Clin. Oral. Implants Res. 25, 266-271.
- 15.Rohrer M D, Schubert C C. (1992) The cutting-grinding technique for histological preparation of undecalcified bone and bone-anchored implants: improvement in instrumentation and procedures. Oral Surg Oral Med Oral Pathol Endod. 74, 73-78.
- 16.Palmquist A, Grandfield K, Norlindh B. (2012) Bone-titanium-oxide interface in humans revealed by transmission electron microscopy and electron. 9, 396-400.
- 17.Iezzi G, Piattelli A, Mangano C. (2014) Peri-implant bone tissues around retrieved human implants after time periods longer than 5 years: a retrospective histologic and histomorphometric evaluation of 8 cases. Odontology. 102-116.
- 18.Mangano C, Piattelli A, Mortellaro C. (2015) Evaluation of peri-implant bone response in implants retrieved for fracture after more than 20 years of loading: a case series. , J. Oral. Implantol 41, 414-418.
- 19.Iezzi G, Piattelli A, Mangano C. (2016) Periimplant bone response in human-retrieved, clinically stable, successful, and functioning dental implants after a long-term loading period: a report of 17 cases from 4 to 20 years. Implant Dent. 25, 380-386.
- 20.Botticello D, Perrotti V, Piattelli A. (2019) Four stable and functioning dental implants retrieved for fracture after 14 and 17 years from the same patient: A histological and histomorphometric. 39, 83-88.
- 21.Vantaggiato G, Iezzi G, Fiera E. (2008) Histologic and histomorphometric report of three immediately loaded screw implants retrieved from man after a three-year loading period. Implant Dent. 17, 192-199.
- 22.Piattelli A, Scarano M, Corigliano M. (1996) Comparison of bone regeneration with the use of mineralized and demineralized freeze-dried bone allografts: a histological and histochemical study in. 17, 1127-31.
- 23.Molly L, Vandromme H, Quirynen M. (2008) Bone formation following implantation of bone biomaterials into extraction sites. J Periodontol. 79, 1108-15.
- 24.Marco D, Luciano A, Corrado R. (2007) Microvessel density in sinus augmentation procedures using anorganic bovine bone and autologous bone: 3 months results. J Implant Dent. 16, 317-25.
- 25.Brunel G, Armand S, Miller N. (2020) Histologic analysis of fractured implant: a case report. , Dent, Int J Periodontics Restorative 20, 521-526.
- 26.Faeda R S, do Nascimento SCS, Santos P L. (2019) Human non-decalcified histology of three dental implants 45 months under function- a case report. , Dent, Int J Implant 5, 10-1186.
- 27.Hudieb M, AlKhader M, Mortaja S. (2021) Impact of bone augmentation of facial bone defect around osseointegrated implant: a three-dimensional finite element analysis. , Dent. J 9, 114-122.
- 28.Piattelli A, Degidi M, Marchetti C. (1997) Histology of the interface of titanium implant retrieved from a nonvascularized mandibular block graft after a 10-month loading period. Int J Maxillofac Implants. 12, 840-843.
- 29.Traini T, Degidi M, Lezzi G. (2007) Comparative evaluation of the peri-implant bone tissue mineral density around unloaded titanium dental implants. J Dent. 35, 84-92.
