Prevalence of Sexually Transmitted Infections in the United Kingdom from Home-Collected Samples: A Cross-Sectional Laboratory Analysis of over 6000 Cases
Abstract
Background
Sexually transmitted infections (STIs) continue to rise globally, with >1 million new cases reported daily in 2020. In England, newly diagnosed STIs increased by 23.8% in 2022 compared to 2021. Many infections remain asymptomatic yet contribute to infertility, pregnancy complications, and neonatal morbidity. While routine screening often focuses on Chlamydia trachomatis, broader detection is limited by laboratory turnaround times and restricted test panels.
Methods
We analysed 6003 home-collected urine and/or swab samples submitted for sexual health screening in the UK. Samples were tested in the laboratory for 10 bacterial and viral pathogens. A total of 5859 urine and 1627 swab samples were processed, with paired samples assessed for diagnostic agreement.
Results
The most common infections detected in urine were Ureaplasma urealyticum (12.1%), Mycoplasma hominis (8.6%), and Chlamydia trachomatis (2.4%). Swabs showed similar prevalence, with Ureaplasma urealyticum (11.6%) most frequent, followed by Mycoplasma hominis (10.4%) and HSV-2 (4.4%). Paired urine–swab samples demonstrated strong agreement, though swabs improved HSV detection.
Conclusions
Ureaplasma urealyticum was the most prevalent STI detected, yet only Chlamydia trachomatis is routinely screened in England. Comprehensive laboratory testing of home-collected samples could reduce the hidden burden of STIs, infertility, pregnancy complications, and neonatal infections, while offering confidential and accessible diagnostics.
Author Contributions
Academic Editor: Ian James Martins, Principal Research Fellow, Edith Cowan University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2026 Mary Jo Kurth, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
MJK, JW, KC, LM, JVL and MWR are employees of Randox Laboratories Ltd but hold no shares in the company. GS is a Medical Director for Randox Health and holds no shares. PF is the Managing Director and owner of Randox Laboratories Ltd.
Citation:
Introduction
Sexually transmitted infections (STIs), and the subset of STIs referred to as sexually transmitted diseases (STDs), are infections/diseases (e.g., chlamydia, gonorrhoea, syphilis) that are spread by sexual activity. Sexual contact can transfer more than 30 different bacteria, viruses and parasites. The most common STIs are caused by eight pathogens, four of which are bacteria (Treponema pallidum causing syphilis, Neisseria gonorrhoeae causing gonorrhoea, Chlamydia trachomatis causing chlamydia and Trichomonas vaginalis causingtrichomoniasis), and four are viral (hepatitis B virus causing hepatitis B, herpes simplex virus (HSV) causing herpes, human immunodeficiency virus (HIV) causing acquired immunodeficiency syndrome (AIDS) and human papillomavirus (HPV), which is linked to cervical cancer). An STI caused by a bacterium is usually treated using antibiotics, for example, chlamydia can be treated with Zithromax (azithromycin) or Vibramycin (doxycycline), and gonorrhoea can be treated with Rocephin (ceftriaxone) or, if a person is allergic to Rocephin, Gentak (gentamicin) plus azithromycin, are usually prescribed. An STI caused by a virus is usually treated with an antiviral or antiretroviral medication (herpes e.g., acyclovir, famciclovir, valacyclovir; HPV e.g., podofilox, imiquimod, sine-catechins). Most viral infections can be managed (e.g., genital warts or cervical dysplasia) with medication.
According to the World Health Organisation (WHO) there were more than 1 million STIs acquired every day in 2020 1. The estimated total number of new infections was 374 million of which 1 in 4 were chlamydia (129 million) 1. In England, 2,195,909 sexual health screens were performed in 2022 by the National Health Service (NHS) (an increase of 13.4% compared to 2021) and there was an increase of 23.8% of newly diagnosed STIs (317,022) compared to 2021 1. The greatest increase in newly diagnosed STIs (50.3%) was for gonorrhoea 1. Disease burden by region is described independently by each public health agency 2, 3, 4. Compared to the ten neighbouring countries in Northern Europe, the UK had the highest number of reported cases of gonorrhoea, fourth highest number of cases for chlamydia, and second highest number of cases for syphilis 5. In the US, one in five individuals (20%) are reported to have an STI (just under 68 million infections) 6.
STIs are spread by unprotected vaginal, oral, and anal sex; often vaginal and rectal infections have no symptoms. Untreated STIs can cause infertility, pelvic inflammatory disease (PID) and can be passed onto a child during vaginal birth. For example, genital herpes caused by HSV infection in pregnant women can cause neonatal HSV in neonates infected during vaginal birth. In the neonates, the infection can affect skin, eyes and/or mouth, and the central nervous system, and mortality is high, if left untreated. Chlamydia trachomatis is also usually asymptomatic in women and if untreated is associated with PID, ectopic pregnancy, chronic pelvic pain and tubal infertility and is also associated with preterm birth, stillbirth, low birth weight and neonatal infections e.g., pneumonia and conjunctivitis 7, 8, 9.
Syphilis infection during pregnancy has also been associated with neonatal infections, in addition to low birth weight, and preterm delivery. Furthermore, syphilis infection during pregnancy is the second leading cause of stillbirth globally 10.
STIs can have a direct impact on sexual and reproductive health through infertility, cancers and pregnancy complications and can also increase the risk of HIV. The natural inflammatory response to an STI produces both CD4 T-cells (“helper” cells) and CD8 T-cells (cytotoxic cells). The CD4 T-cells direct the CD8 T-cells, to neutralise the STI infection. However, CD4 T-cells are potential targets for HIV infection 11, 12. Therefore, the more effective the inflammatory response, the more CD4 T-cells that are recruited, improving the likelihood of HIV penetrating the immune defences 11, 12. Thus, early identification and/or screening for STIs, especially in asymptomatic individuals is important in reducing potential transmission. Currently there are no tests that have the sensitivity or specificity to be 100% accurate. However, the nucleic acid amplification test (NAAT) is the current standard of care for all cases, including extra-genital infections and medico-legal cases 13, 14, 15, 16. The testing guidelines for STIs in the UK are prepared by the British Association for Sexual Health and HIV (BASHH) and the National Institute for Clinical Excellence (NICE) (Supplementary 1 and 2, respectively) 17, 18.
Laboratory tests often have long turnaround times, which can delay effective treatment, and potentially lead to an increase in transmission and can contribute to an individual’s anxiety. Confidante®, a home STI test kit enables an individual to collect a urine and/or swab sample in the privacy of their own home and receive their sexual health results confidentially. Following COVID, there has been greater acceptance for the use of home testing kits in diagnostics. For example, in 2020, the number of home testing kits issued by the NHS (each kit tests for chlamydia, gonorrhoea, syphilis and HIV) increased by 61% compared to 2019. Almost 60% of the tests issued were to individuals aged between 20-29 years 19.
The availability of home STI test kits, such as Confidante®, tests for Chlamydia trachomatis, Neisseria gonorrhoea, HPV1, HPV2, Treponema pallidum, Mycoplasma hominis, Ureaplasma urealyticum, Mycoplasma genitalium, Haemophilus ducreyi and, Trichomonas vaginalis, is enabling key population groups and individuals, who may not want, or be able to attend a face-to-face service for several reasons, including age, socioeconomic circumstances, ethnicity and/or geographic location. Non-NHS home STI test kits are not reimbursed, however, without home STI test kits, these individuals may not self-test and as a result develop significant morbidity and could potentially contribute to onward transmission of STIs.
A blood test for 3 infectious diseases (hepatitis B, HIV, and syphilis) is offered to women during pregnancy as part of their routine antenatal screening, which is recommended for every pregnancy. Other STI ‘at risk groups’ include men who have sex with men and other sexual and gender diverse individuals (e.g., transgender women), adolescents and young adults, and people of colour 20.
The aim of this study was to analyse the incidence of STI infection in UK individuals who submitted a urine and/or swab sample for investigation from 3rd June 2014 to 21st November 2022 (Supplementary 3), using the Confidante® kit (Randox Laboratories Ltd, Crumlin, UK) to report the number of positive infections detected from urine and/or swab samples provided by the individual. In addition, the distribution of infection by age group and the number of infections per sample were also reviewed.
Materials and Methods
Data was collected from n=6003 UK individuals (total number of individuals) who had submitted a urine and/or swab sample for sexual health screening using the Confidante® STI kit (Figure 1). Client data was anonymous. Demographics included age and gender. In total, results from n=5859 urine samples and n=1627 swab samples from n=6003 individuals were included in the study (n=1304 individuals provided both a urine and a swab sample “paired”). Consent was obtained digitally through Randox’s online booking platforms.
Figure 1.Confidante® STI testing kit
Confidante® Home STI Testing Kit
Confidante® is a home testing kit that allows an individual to collect a urine and/or swab sample (s) and post to Randox Laboratories Ltd for screening of 10 sexually transmitted infections: Chlamydia trachomatis, Neisseria gonorrhoea, herpes simplexvirus1, herpes simplexvirus2, Treponema pallidum, Mycoplasma hominis, Ureaplasma urealyticum, Mycoplasma genitalium, Haemophilus ducreyi and, Trichomonas vaginalis. Individuals can purchase a Randox Confidante® STI test kit online, at their local pharmacy, or at Randox Health Clinics throughout the UK. Easy-to-follow instructions, provided within the Confidante® kit, direct the individual how to collect their sample (s). For individuals that are asymptomatic, a urine sample is required; for those that are symptomatic (i.e., showing signs of infection e.g., sores, painful or burning urination and/or unusual discharge), a urine sample and swab (of the infected area) are required. Individuals register their Confidante® kit online (confidential), post their sample(s) to Randox Laboratories Ltd, and receive their results electronically in less than one week. All tests are performed using biochip array technology (which incorporates PCR-based technology and hybridisation to a biochip (sensitivity and specificity data is included in Table 1) 21 at Randox’s ISO17025 accredited laboratory (Randox Clinical Laboratory Services (RCLS), Randox Science Park, 30 Randalstown Road, Antrim, BT41 4FL, UK) on an Evidence Investigator instrument (Randox Laboratories Ltd).
Table 1. Randox STI PCR sensitivity and specificity data| TP | FP | TN | FN | Total Samples | Concordance (%) | Sensitivity (%) | Specificity (%) | Chi- Sq Sig | Fisher's Sig | PPV (%) | NPV (%) | Prevalence (%) | |
| CT | 37 | 0 | 735 | 0 | 772 | 100 | 100 | 100 | 0.0001 | 0.001 | 100 | 100 | 4.8 |
| NG | 8 | 3 | 761 | 0 | 772 | 100 | 100 | 100 | 0.0001 | 0.001 | 73 | 100 | 1.0 |
| HSV1 | 69 | 10 | 229 | 0 | 308 | 97 | 100 | 96 | 0.0001 | 0.001 | 87 | 100 | 22.4 |
| HSV2 | 42 | 17 | 250 | 0 | 309 | 94 | 100 | 94 | 0.0001 | 0.001 | 71 | 100 | 13.6 |
| TP | 11 | 0 | 243 | 0 | 254 | 100 | 100 | 100 | 0.0001 | 0.001 | 100 | 100 | 4.3 |
| TV | 5 | 3 | 175 | 0 | 183 | 98 | 100 | 98 | 0.0001 | 0.001 | 63 | 100 | 2.7 |
| MH | 71 | 20 | 287 | 0 | 378 | 95 | 100 | 93 | 0.0001 | 0.001 | 78 | 100 | 18.8 |
| MG | 12 | 4 | 338 | 1 | 355 | 99 | 92 | 99 | 0.0001 | 0.001 | 75 | 100 | 3.4 |
| UU | 110 | 15 | 247 | 0 | 372 | 96 | 100 | 94 | 0.0001 | 0.001 | 88 | 100 | 29.6 |
Statistical Analysis
Statistical analysis was performed using R 22. The association between swab positive and urine positive results and swab negative and urine negative results was assessed using a matrix of results and Cramer’s V output where Cramer’s V was used to calculate the correlation between nominal categorical variables and a value >0.7 was considered a strong correlation. Data visualisations were generated using R.
Results
Demographics
Individuals (n=6003) registered their Confidante® STI kit online, at which time they also provided their age and gender. Individuals were aged between 16-75 years with a median age of 33 years. There was n=3405/6003 (56.7%) males, n=2110/6003 (35.1%) females and n=488/6003 (8.1%) with no recorded gender. Of the n=1355 individuals that tested positive for any of the measured STIs, from either urine or swab samples, n=538/1355 (43.4%) were male, n=703/1355 (51.9%) were female, and n=114/1355 (8.4%) did not report gender (“Unknown”). There were more Chlamydia trachomatis infections in males, whereas there was more Mycoplasma genitalium, Mycoplasma hominis and Ureaplasma urealyticum infections in females (Table 2).
Table 2. Number of infections detected in urine and swab by gender, and unknown| Male n=538 n (%) | Female n=703 n (%) | Unknown n=114 n (%) | ||
| Chlamydia trachomatis | URINE | 72 (13.38%) | 55 (7.82%) | 12 (10.53%) |
| SWAB | 12 (2.23%) | 19 (2.7%) | 6 (5.26%) | |
| Haemophilus ducreyi | URINE | 1 (0.19%) | 0 (0%) | 0 (0%) |
| SWAB | 0 (0%) | 0 (0%) | 0 (0%) | |
| Herpes simplexvirus1 | URINE | 15 (2.79%) | 11 (1.56%) | 8 (7.02%) |
| SWAB | 18 (3.35%) | 18 (2.56%) | 6 (5.26%) | |
| Herpes simplexvirus2 | URINE | 18 (3.35%) | 30 (4.27%) | 7 (6.14%) |
| SWAB | 21 (3.9%) | 37 (5.26%) | 9 (7.89%) | |
| Mycoplasma genitalium | URINE | 42 (7.81%) | 38 (5.41%) | 10 (8.77%) |
| SWAB | 10 (1.86%) | 20 (2.84%) | 11 (9.65%) | |
| Mycoplasma hominis | URINE | 150 (27.88%) | 337 (47.94%) | 11 (9.65%) |
| SWAB | 18 (3.35%) | 126 (17.92%) | 17 (14.91%) | |
| Neisseria gonorrhoea | URINE | 8 (1.49%) | 6 (0.85%) | 7 (6.14%) |
| SWAB | 8 (1.49%) | 3 (0.43%) | 5 (4.39%) | |
| Treponema pallidum | URINE | 1 (0.19%) | 0 (0%) | 0 (0%) |
| SWAB | 2 (0.37%) | 0 (0%) | 0 (0%) | |
| Trichomonas vaginalis | URINE | 2 (0.37%) | 8 (1.14%) | 13 (11.4%) |
| SWAB | 6 (1.12%) | 8 (1.14%) | 17 (14.91%) | |
| Ureaplasma urealyticum | URINE | 294 (54.65%) | 380 (54.05%) | 29 (25.44%) |
| SWAB | 43 (7.99%) | 116 (16.5%) | 22 (19.3%) |
Urine sample results
The two most common infections detected in urine using the Confidante® kit were Ureaplasma urealyticum and Mycoplasma hominis (Table 3 and Figure 2).
A total of n=4608/5859 (78.6%) urine samples tested negative for any STI infection, whereas n=954/5859 (16.3%) tested positive for one STI infection, n=270/5859 (4.6%) for 2 STI infections, n=25/5859 (0.4%) for 3 STI infections and n=2/5859 (<0.1%) for 4 STI infections.
Figure 2.Number of positive results for each STI infection detected from urine and swab samples. The two most common STI infections detected in urine using the Confidante® kit were Ureaplasma urealyticum (n=707 urine, n=188 swab) and Mycoplasma hominis (n=503 urine, n=169 swab).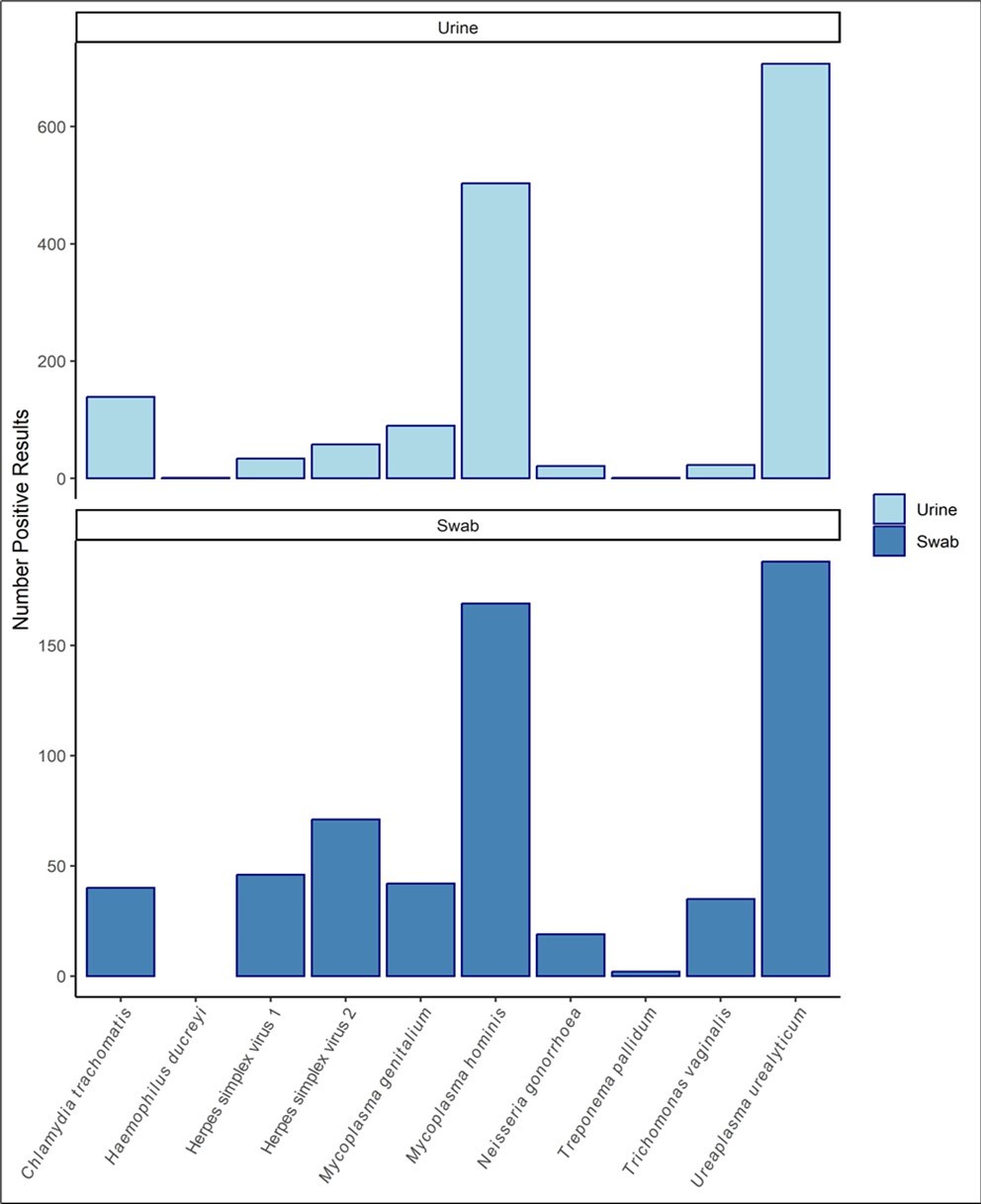
| Urine | Swab | ||
| # | Infectious agent | Positives/total (%) | Positives/total (%) |
| 1 | Ureaplasma urealyticum | 707/5859 (12.1) | 188/1627 (11.6) |
| 2 | Mycoplasma hominis | 503/5859 (8.6) | 169/1627 (10.4) |
| 3 | Chlamydia trachomatis | 139/5859 (2.4) | 40/1627 (2.5) |
| 4 | Mycoplasma genitalium | 90/5859 (1.5) | 42/1627 (2.6) |
| 5 | Herpes simplexvirus2 | 58/5859 (1.0) | 71/1627 (4.4) |
| 6 | Herpes simplexvirus1 | 34/5859 (0.6) | 46/1627 (2.8) |
| 7 | Trichomonas vaginalis | 23/5859 (0.4) | 35/1627 (2.2) |
| 8 | Neisseria gonorrhoea | 21/5859 (0.4) | 19/1627 (1.2) |
| 9 | Treponema pallidum | 1/5859 (<0.1) | 2/1627 (<0.1) |
| 10 | Haemophilus ducreyi | 1/5859 (<0.1) | 0/1627 (0) |
Swab sample results
The two most common STI infections detected from swab samples were Ureaplasma urealyticum and Mycoplasma hominis (Table 3). A total of n=1175/1627 (72.2%) swab samples tested negative for any STI infection whereas, n=316 tested positive for 1 STI infection (19.4%), n=114 (7.0%) for 2 STI infections, n=20 (1.2%) for 3 STI infections, and, n=2 (0.1%) for 4 STI infections.
Comparison of swab and urine results
According to Confidante® kit instructions, individuals that are asymptomatic should provide a urine sample; for those that are symptomatic (i.e., showing signs of infection), a urine sample and swab of the infected area, are required. A total of n=1396/6003 (23.3%) individuals provided both urine and swab samples. Where both urine and swab samples were tested from the same individual (paired sample), there was strong agreement between STI infections detected from both matrices as shown by the Cramer’s V statistic (Table 4). Although HSV1 was detected in n=37 swabs, n=17 of these positive swab samples had a negative result for HSV1 for the associated urine sample. However, when the swab was negative, the associated urine sample was also negative for HSV1. Similarly, for HSV2, of the n=56 swabs that were positive, n=18 of the associated urine samples were negative for HSV2. These results demonstrate the need to include a swab sample for the accurate detection of HSV1 and 2. Of the n=1396 paired urine and swab samples, n=40 paired samples for the detection of Ureaplasma urealyticum did not agree, and n=33 paired samples for the detection of Mycoplasma hominis also did not agree.
There was a strong association between swab positive and urine positive results and swab negative and urine negative results, as demonstrated by high (>0.7) Cramer’s V output (Table 4). Results for Haemophilus ducreyi, from pairedurine and serum samples were negative, therefore, Cramer’s V was not calculated.
Table 4. Comparison matrix of test results when a urine and swab sample were provided from the same individual (paired sample) (n=1384). Note, not every individual had a positive urine and a positive swab result.| URINE | SWAB | Cramer's V | ||
| Negative | Positive | |||
| Chlamydia trachomatis | Negative | 1359 | 6 | 0.891 |
| Positive | 2 | 29 | ||
| Haemophilus ducreyi | Negative | 1396 | 0 | - |
| Positive | 0 | 0 | ||
| Herpes simplex virus 1 | Negative | 1359 | 17 | 0.741 |
| Positive | 0 | 20 | ||
| Herpes simplex virus 2 | Negative | 1339 | 18 | 0.803 |
| Positive | 1 | 38 | ||
| Mycoplasma genitalium | Negative | 1359 | 6 | 0.841 |
| Positive | 4 | 27 | ||
| Mycoplasma hominis | Negative | 1236 | 13 | 0.872 |
| Positive | 20 | 127 | ||
| Neisseria gonorrhoea | Negative | 1381 | 5 | 0.768 |
| Positive | 1 | 9 | ||
| Treponema pallidum | Negative | 1394 | 1 | 0.707 |
| Positive | 0 | 1 | ||
| Trichomonas vaginalis | Negative | 1375 | 8 | 0.825 |
| Positive | 0 | 13 | ||
| Ureaplasma urealyticum | Negative | 1211 | 10 | 0.865 |
| Positive | 30 | 145 | ||
STI Infection classification by age group
To determine the STI infection classification by age group, individuals were categorised by age, sample type and STI infection. STI infection by age and classification is shown in Figure 3. There was n=1355 individuals positive for any of the measured STIs in either urine or swab. The age group with the highest incidence of any STI infection was 20-29 years (n=449/1355, 33.1%). The 30-39 year age group had the second highest incidence of any STI infection (n=328/1355, 24.2%).
Figure 3.Number of urine and swab samples positive for STI infection by age group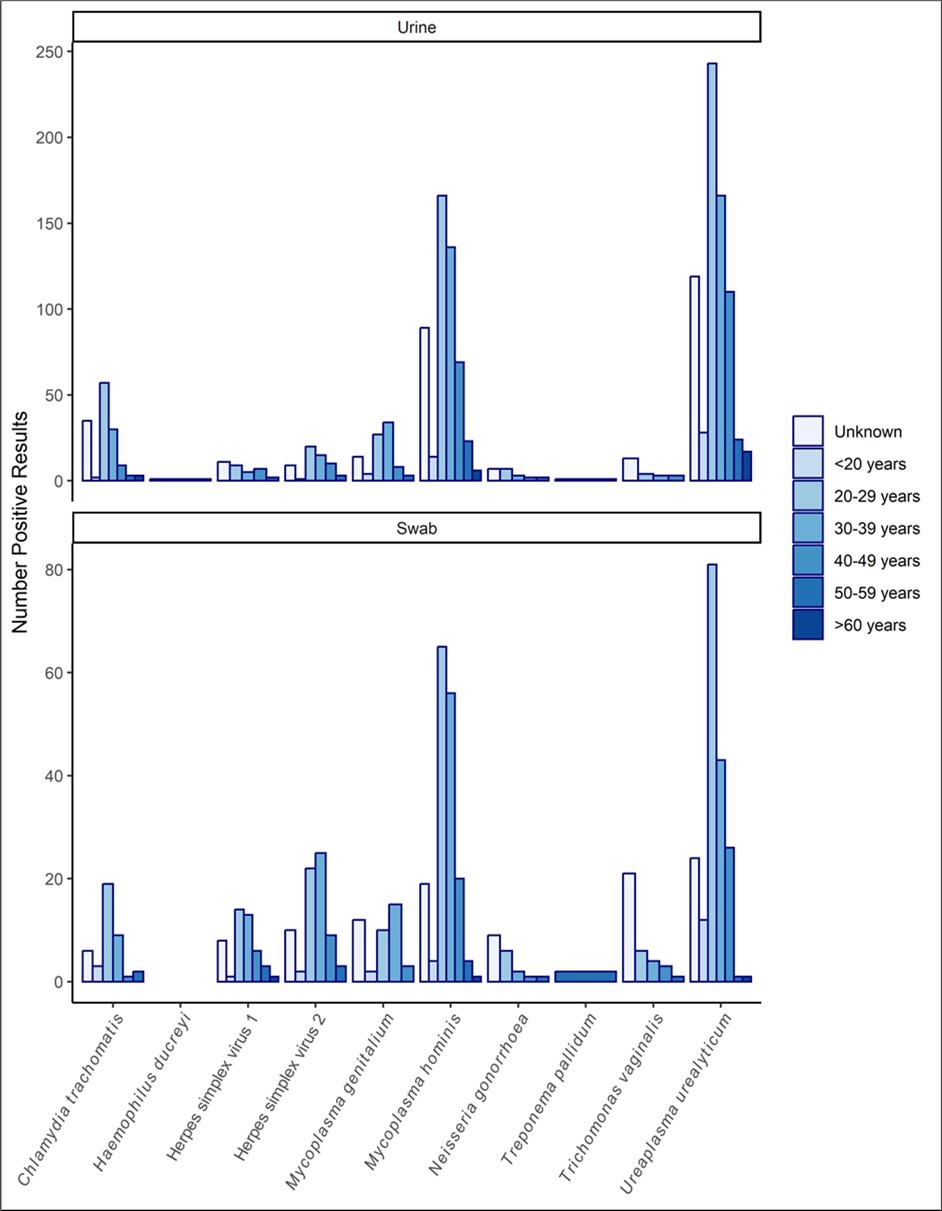
Discussion
According to the World Health Organisation (WHO) there were more than 1 million STIs reported every day in 2020 1. The estimated total number of new STI infections was 374 million of which 1 in 4 were chlamydia (129 million) 1. Some STIs have no symptoms, however, STIs can have a direct impact on sexual and reproductive health causing infertility, cancers, pregnancy complications, and an increased risk of HIV 11, 12, 23.
The aim of this work was to report the STI results from UK individuals that provided a urine and/or swab sample between 3rd June 2014 to 21st November 2022, using the Randox Confidante® kit (Randox Laboratories Ltd, Crumlin, UK) to identify the numbers of positive infections detected from urine and/or swab samples. In addition, the distribution of STI infection by age group and the number of STI infections per sample were also reported. The Confidante® STI kit allows an individual to collect a sample at home, which is then posted to Randox Laboratories to test for ten STIs.
The length of time from an STI infection (exposure) to symptoms is known as the window period (WP). However, the exposure time of testing may not be within the WP. For chlamydia and gonorrhoea STI infection, the WP is 2 weeks; HIV WP 7 weeks; syphilis, hepatitis B and C, the WP 12 weeks 24. However, sometimes symptoms may not appear until months, or even years later. This may potentially explain the results observed in the current study where of the n=5859 urine samples that were tested, 78.6% tested negative, and of the n=1627 swab samples that were tested, almost 72.2% tested negative. This would also suggest there is significant merit in repeat testing at different time periods e.g., at the WP, or after a change of sexual partner.
Of the samples that were positive for STI infection, most had only 1 STI infection (16.3% for urine and 19.4% for swabs). The most common STI infective agent in urine samples was Ureaplasma urealyticum followed by Mycoplasma hominis, HSV2, HSV1, Mycoplasmagenitalium, Chlamydiatrachomatis, Trichomonas vaginalis, Neisseria gonorrhoea, Treponema pallidum, and Haemophilus ducreyi. It is worth noting that where a urine and swab sample were provided by the same individual, the results did not always match (Table 4). For example, a possible explanation is that the HSV1 and 2 infections cause a sore which should be swabbed directly. If the sore caused by HSV1 and HSV2 is not in the path of urine, the virus may not be shed into the urine sample and, therefore, not detected. Moreover, the swab test is usually taken from an affected area, such as a blister or a sore.
Ureaplasma urealyticum is very contagious infection that is commonly found in the genital flora of sexually active individuals. Ureaplasma urealyticum often has no symptoms, and a large percentage of infected individuals never experience any problems. However, if left undetected, and untreated, Ureaplasma urealyticum can lead to infertility, highlighting the need for regular sexual health checks.
Mycoplasma hominis affects up to 50% of sexually active individuals. The bacterium is commonly found in the urinary tract in small quantities however, at high levels the bacterium can cause infection, which can be transmitted sexually. In the US there are no screening or treatment recommendations for Ureaplasmaspecies or Mycoplasma hominis. Mycoplasma genitalium testing is only recommended for males with persistent urethritis, or females with cervicitis or PID 25. Interestingly, motility of sperm is decreased following adhesion of Ureaplasmaurealyticum26 and the byproducts are also toxic to sperm 27 causing sperm to be susceptible to peroxidation damage, which can result in infertility 28. However, other studies reported no effect on male fertility 29. There is some speculation that the differences observed in the studies may be related to the time, or level of infection, and possibly infection by multiple bacteria 30. Similarly, Mycoplasma can infect the male reproductive system. Mycoplasma hominis is associated with low sperm count and motility 31. Mycoplasma gentialiummay not be associated with male infertility however, it can impair female fertility if transmitted during sex 32.
Chlamydia trachomatis, an intracellular bacterium, can also affect male fertility by infecting testicular cells (sperm, Leydig and Sertoli) within 3 days of exposure 33, resulting in reduced sperm quality 34. Chlamydiatrachomatis, usually asymptomatic in females, if untreated can result in reproductive damage, and is associated with tubal infertility, PID, chronic pelvic pain and ectopic pregnancy 23. Preterm birth, stillbirth, low birth weight and neonatal infections e.g. conjunctivitis and pneumonia, can occur as a result of Chlamydiatrachomatis infection in the mother during pregnancy 7, 8, 9. Early diagnosis and treatment of Chlamydiatrachomatis are critical to preventing these conditions.
Routine screening for STIs often include testing for chlamydia, gonorrhoeaand HPV, however, from the results presented in this analysis, it is evident that although these infections were detected, the three most common infections were Ureaplasma urealyticum, Myoplasma hominis and Chlamydia trachomatis. If untreated, Ureaplasma urealyticum, Myoplasma hominis and Chlamydia trachomatis can affect fertility, which may impact future pregnancy 23, 26, 32. In the UK, 53,000 patients undertook 69,000 fresh and frozen IVF cycles and 5,700 donor insemination cycles in 2019. According to NICE (National Health Service (NHS) UK guidelines) up to 3 cycles of IVF should be offered on the NHS but some Integrated Care Boards only offer 1 cycle 35. Indeed, one cycle of treatment can cost over £5,000 35. This highlights the clinical utility for repeat STI screening.
Where both urine and swab samples were tested from the same individual (paired samples), there was strong agreement between STI infections detected from both matrices as shown by the Cramer’s V statistic. However, the results also demonstrate the need to include a swab sample for the accurate detection of HSV1 and 2.
Unsurprisingly, individuals in the 20-29 age group had the highest reported incidence of STIs. This was followed by 30-39 year olds. In the US, women <25 years account for most STI infections, therefore, annual screening in this age group is recommended to reduce the frequency of PID and other adverse health outcomes and potential complications 7, 36, 37. Similarly, in the UK and Europe, the impact of STIs remains greatest in individuals aged 15 to 24 years 1, 38. The Randox Confidante® STI test detects the presence of 10 infectious agents. The 10 results are obtained from a urine and/or a swab sample, which can be collected at home using the Confidante® collection kit. The results reported from the STI test provide data to the individual to share with their clinician improving evidence-based decision-making, antibiotic stewardship and patient outcome. Clinical utility of the Confidante® STI kit has the potential to reduce the current and future burden of STIs on the healthcare system, reduce infertility and IVF treatments and reduce complications during pregnancy and neonate infections. The Confidante® STI test (CE marked) provides an individual the opportunity to screen their own sexual health in the privacy of their own home.
Study Limitations
The results showed that not all individuals that provided samples for STI screening, recorded information on their gender and age. HIV, hepatitis, and other blood borne STIs were not considered within this report. The time from potential infection to testing is not recorded for each sample, thus the WP of infection is unknown. Individuals collect their own samples, and it is possible that the swab/urine may be used incorrectly or potentially contaminated. This study was limited to those who purchased the test kit, which limits the generalisability of the results.
Authors Contribution
MJK wrote the manuscript and MWR, KC, LM, JL, GS and PF assisted with reviewing and editing the manuscript. JW compiled the data, undertook the analyses, and produced the figures.
Supplementary Material
References
- 1. (2023) Sexually transmitted infections and screening for chlamydia in England-2022 report - GOV.UK [Internet]. Available from: https://www.gov.uk/government/statistics/sexually-transmitted-infections-stis-annual-data-tables/sexually-transmitted-infections-and-screening-for-chlamydia-in-england-2022-report.
- 2. (2024) Sexually transmitted infections | HSC Public Health Agency [Internet]. Available from: https://www.publichealth.hscni.net/directorate-public-health/health-protection/sexually-transmitted-infections.
- 3. (2024) Sexually transmitted infections - ScotPHO [Internet]. Available from: https://www.scotpho.org.uk/risk-factors/sexual-health/data/sexually-transmitted-infections/.
- 4. (2023) Sexual Health in Wales: Sexually Transmitted Infections, Emergency and Long-acting Reversible Contraception provision and Termination of Pregnancy Annual report.
- 5. (2024) Surveillance Atlas of Infectious Diseases [Internet]. Available from: https://www.ecdc.europa.eu/en/surveillance-atlas-infectious-diseases.
- 6. (2023) STI Prevalence, Incidence, and Cost Estimates [Internet]. Available from: https://www.cdc.gov/std/statistics/prevalence-incidence-cost-2020.htm.
- 7.He W, Jin Y, Zhu H, Zheng Y, Qian J. (2020) Effect of Chlamydia trachomatis on adverse pregnancy outcomes: a meta-analysis. Arch Gynecol Obstet. 302(3), 553-67.
- 8.Hammerschlag M R, Chandler J W, Russell Alexander E, English M, Koutsky L.Longitudinal studies on chlamydial infections in the first year of life. Pediatr Infect Dis. 1(6), 395-401.
- 9.Hammerschlag M R. (2011) Chlamydial and gonococcal infections in infants and children. Clin Infect Dis. , Suppl 3.
- 10. (2023) Data on syphilis [Internet]. Available from: https://www.who.int/data/gho/data/themes/topics/topic-details/GHO/data-on-syphilis.
- 11.Looker K J, JAR Elmes, Gottlieb S L, Schiffer J T, Vickerman P et al. (2017) Effect of HSV-2 infection on subsequent HIV acquisition: an updated systematic review and meta-analysis. Lancet Infect Dis. 17(12), 1303.
- 12.Jarvis G A, Chang T L. (2012) Modulation of HIV Transmission by Neisseria gonorrhoeae: Molecular and Immunological Aspects. Curr HIV Res. 10(3), 211.
- 13.Skidmore S, Horner P, Mallinson H. (2006) Testing specimens for Chlamydia trachomatis. Sex Transm Infect. 82(4), 272-5.
- 15.K V Ota, Tamari I E, Smieja M, Jamieson F, Jones K E et al. (2009) Detection of Neisseria gonorrhoeae and Chlamydia trachomatis in pharyngeal and rectal specimens using the BD Probetec ET system, the Gen-Probe Aptima Combo 2 assay and culture. Sex Transm Infect. 85(3), 182-6.
- 16.Alexander S, Ison C, Parry J, Llewellyn C, Wayal S et al. (2008) Self-taken pharyngeal and rectal swabs are appropriate for the detection of Chlamydia trachomatis and Neisseria gonorrhoeae in asymptomatic men who have sex with men. Sex Transm Infect. 84(6), 488-92.
- 17.Coleman H, Samuel I, Soni S, Murchie M, Clarke M et al. (2023) BASHH Summary Guidance on Testing for Sexually Transmitted Infections.
- 19. (2024) New STI stats reveal a move to at home testing | HSC Public Health Agency [Internet]. Available from: https://www.publichealth.hscni.net/news/new-sti-stats-reveal-move-home-testing.
- 20.Vermund S H, Geller A B, Crowley J S. (2021) Sexually Transmitted Infections: Adopting a Sexual Health Paradigm. Sex Transm Infect.
- 21.Molloy R M, Mc Connell RI, Lamont J V, FitzGerald S P. (2005) Automation of Biochip Array Technology for Quality Results. Clin Chem Lab Med. 43(12), 1303-13.
- 22.R Core Team.R core team (2021). R: A language and environment for statistical computing. R Foundation for Statistical Computing. , Vienna, Austria. URL http://www.R-project.org
- 23.Van Gerwen OT, Muzny C A, Marrazzo J M. (2022) Sexually transmitted infections and female reproductive health. Nat Microbiol. 7(8), 1116-26.
- 24.Wang L, Shao C, Lu L, Liu J, Yang Z et al. (2021) A Longitudinal Case Study of Concurrent Infection with Syphilis and Human Immunodeficiency Virus During the Early Phase. AIDS Res Hum Retroviruses. 37(7), 523-8.
- 25.Manhart L E, Broad J M, Golden M R. (2011) Mycoplasma genitalium: should we treat and how? Clin Infect Dis. , Suppl 3.
- 26.Núñez-Calonge R, Caballero P, Redondo C, Baquero F, Martínez-Ferrer M et al. (1998) Ureaplasma urealyticum reduces motility and induces membrane alterations in human spermatozoa. Hum Reprod. 13(10), 2756-61.
- 27.Farsimadan M, Motamedifar M. (2020) Bacterial infection of the male reproductive system causing infertility. J Reprod Immunol.
- 28.Shang X J, Huang Y F, Xiong C L, Xu J P, Yin L et al. (1999) Ureaplasma urealyticum infection and apoptosis of spermatogenic cells. , Asian J Androl 1(3), 127-9.
- 29.Gdoura R, Kchaou W, Chaari C, Znazen A, Keskes L et al. (2007) Ureaplasma urealyticum, Ureaplasma parvum, Mycoplasma hominis and Mycoplasma genitalium infections and semen quality of infertile men. BMC Infect Dis. 8, 129.
- 30.Wang S, Zhang K, Yao Y, Li J, Deng S. (2021) Bacterial Infections Affect Male Fertility: A Focus on the Oxidative Stress-Autophagy Axis. Front Cell Dev Biol.
- 31.Lee J S, Kim K T, Lee H S, Yang K M, Seo J T et al. (2013) Concordance of Ureaplasma urealyticum and Mycoplasma hominis in infertile couples: impact on semen parameters. Urology. 81(6), 1219-24.
- 32.Huang C, Zhu H L, Xu K R, Wang S Y, Fan L Q et al. (2015) Mycoplasma and ureaplasma infection and male infertility: a systematic review and meta-analysis. Andrology. 3(5), 809-16.
- 33.Bryan E R, Kollipara A, Trim L K, Armitage C W, Carey A J et al. (2019) Hematogenous dissemination of Chlamydia muridarum from the urethra in macrophages causes testicular infection and sperm DNA damage†. Biol Reprod. 101(4), 748-59.
- 34.Satta A, Stivala A, Garozzo A, Morello A, Perdichizzi A et al.Experimental Chlamydia trachomatis infection causes apoptosis in human sperm. Hum Reprod. 21(1), 134-7.
- 36.Workowski K A, Bachmann L H, Chan P A, Johnston C M, Muzny C A et al. (2021) Sexually Transmitted Infections Treatment Guidelines. 70(4), 1-187.
